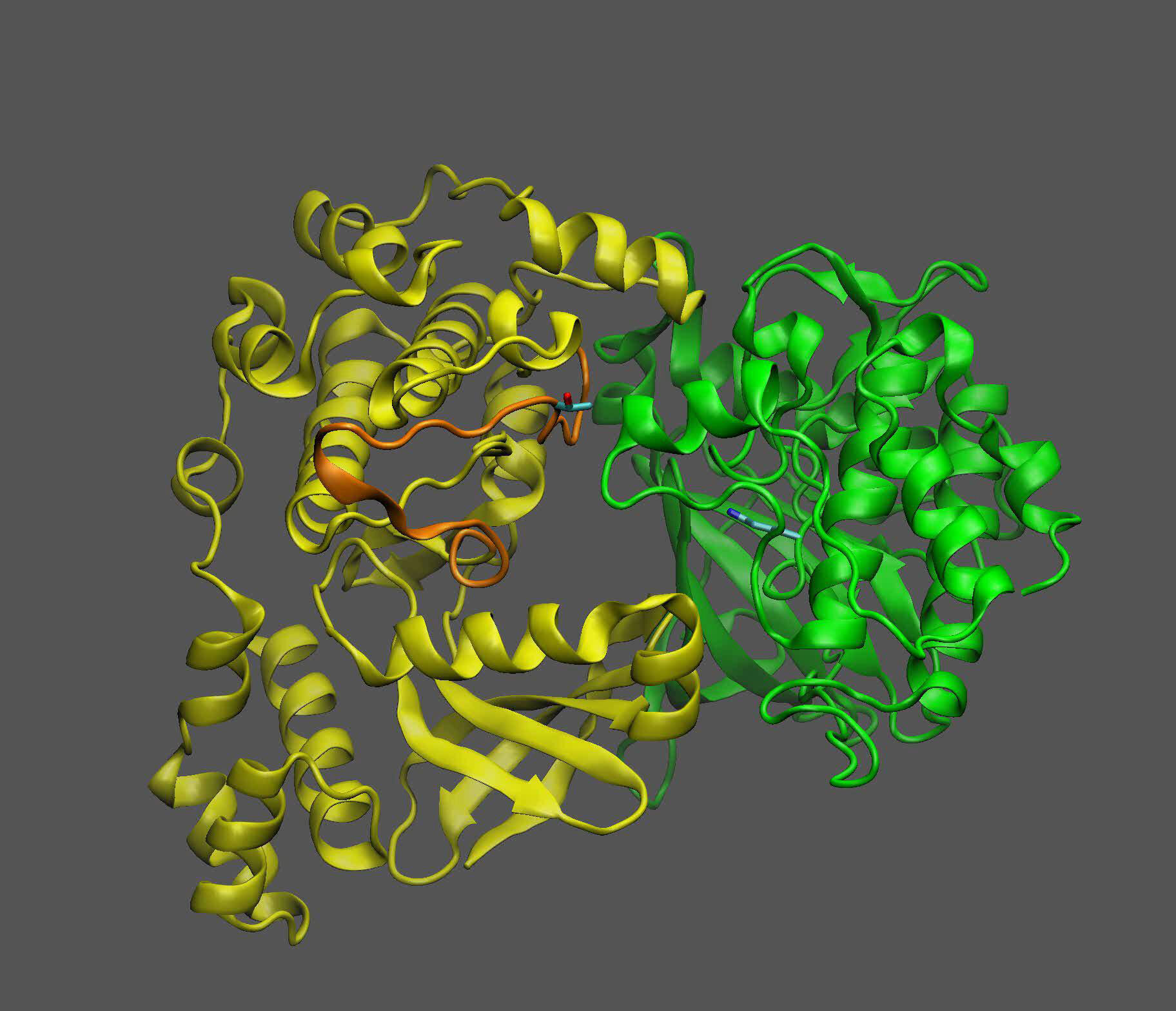
This image shows a plant protein known as KIN10 (yellow), which acts as a sensor and switch to turn oil production off or on depending on whether it interacts with another protein (green). Credit: Brookhaven National Laboratory
× near
This image shows a plant protein known as KIN10 (yellow), which acts as a sensor and switch to turn oil production off or on depending on whether it interacts with another protein (green). Credit: Brookhaven National Laboratory
Proteins are molecular machines with flexible parts and moving parts. Understanding how these parts move helps scientists unravel the function the protein plays in living things — and potentially how to modify its effects. Biochemists at the US Department of Energy’s (DOE’s) Brookhaven National Laboratory and colleagues at the DOE’s Pacific Northwest National Laboratory (PNNL) have published a new example of how such a molecular machine works.
Their journal article Scientific progress describes how the moving parts of a particular plant protein control whether plants can grow and produce energy-intensive products like oil—or instead enact a series of steps to conserve valuable resources. The study focused specifically on how the molecular machinery is regulated by a molecule that rises and falls with the level of sugar, the main energy source of plants.
“This paper reveals the detailed mechanism that tells plant cells ‘we have a lot of sugar,’ and then how this signaling affects biochemical pathways that trigger processes such as plant growth and oil production,” said Brookhaven Lab biochemist Jantana Blanford, head of the the study author.
The study builds on earlier work by the Brookhaven team that revealed the molecular links between sugar levels and oil production in plants. One potential goal of this research is to identify specific proteins — and parts of proteins — scientists can engineer to make plants that produce more oil for use as biofuels or other petroleum-based products.
“Identifying exactly how these molecules and proteins interact, as this new study does, brings us closer to identifying how we can engineer these proteins to increase vegetable oil production,” said John Shanklin, chair of Brookhaven Lab’s biology department and head of the research team.
Revealing molecular interactions
The team used a combination of laboratory experiments and computational modeling to understand how the molecule, which serves as a sugar proxy, binds to a “sensor kinase” known as KIN10.
KIN10 is the protein that contains the moving parts that determine which biochemical pathways are turned on or off. Scientists already knew that KIN10 acts as both a sugar sensor and a switch: when sugar levels are low, KIN10 interacts with another protein to trigger a cascade of reactions that eventually shuts down oil production and breaks down fat-rich energy molecules such as oil and starch to produce energy that powers the cell.
But when sugar levels are high, KIN10’s shut-down function is turned off—meaning plants can grow and produce lots of oil and other products with the abundant energy.
But how does connecting the sugar proxy to the KIN10 flip the switch?
This diagram shows the two pathways KIN10 and its neighboring protein GRIK1 follow under low and high sugar conditions. The low sugar content allows the addition of phosphate (P) to KIN10, which triggers a phosphorylation cascade that leads to the degradation of enzymes involved in oil synthesis. This involves degradation of WRI1, the master switch for oil synthesis. When sugar is abundant, however, a sugar proxy molecule (T6P) binds to the KIN10 chain to block its interaction with GRIK1. This keeps the pathway for oil synthesis open. Credit: Brookhaven National Laboratory
× near
This diagram shows the two pathways KIN10 and its neighboring protein GRIK1 follow under low and high sugar conditions. The low sugar content allows the addition of phosphate (P) to KIN10, which triggers a phosphorylation cascade that leads to the degradation of enzymes involved in oil synthesis. This involves degradation of WRI1, the master switch for oil synthesis. When sugar is abundant, however, a sugar proxy molecule (T6P) binds to the KIN10 chain to block its interaction with GRIK1. This keeps the pathway for oil synthesis open. Credit: Brookhaven National Laboratory
To find out, Blanford started with the saying “opposites attract.” She identified three positively charged parts of KIN10 that could be attracted to the abundant negative charges of the sugar proxy molecule. A laboratory-based process of elimination, which involves making variations of KIN10 with modifications at these sites, identified the only true binding site.
The Brookhaven team then turned to their computational colleagues at PNNL.
Marcel Baer and Simone Raugei of PNNL investigated at the atomic level how the sugar proxy and KIN10 fit together.
“By using multiscale modeling, we observed that the protein can exist in multiple conformations, but only one of them can effectively bind the sugar proxy,” Baer said.
PNNL simulations identify key amino acids in the protein that control sugar binding. These computational insights were then confirmed experimentally.
The combined body of experimental and computational information helped the scientists understand how the interaction with the sugar proxy directly affects KIN10’s downstream action.
Turn the key
“Further analysis showed that the entire KIN10 molecule is rigid, except for one long flexible chain,” Shanklin said. The models also showed that the loop’s flexibility is what allows KIN10 to interact with an activator protein to trigger the cascade of reactions that ultimately shut down oil production and plant growth.
When sugar levels are low and there is little proxy sugar molecule, the chain remains flexible and the shutdown mechanism can operate to reduce plant growth and oil production. That makes sense to conserve valuable resources, Shanklin said.
But when sugar levels are high, the sugar proxy binds tightly to KIN10.
“The calculations show how this small molecule blocks the rotation of the loop and prevents it from triggering the shutdown cascade,” Blanford said.
Again, this makes sense since an abundance of sugar is available to plants for oil production.
Now that scientists have this detailed information, how might they use it?
“Potentially, we could use our new knowledge to engineer KIN10 with altered binding strength for the sugar proxy to change the set point where plants make things like oil and break things down,” Shanklin said.
More info:
Jantana Blanford et al, Molecular mechanism of trehalose 6-phosphate inhibition of the plant metabolic sensor kinase SnRK1, Scientific progress (2024). DOI: 10.1126/sciadv.adn0895. www.science.org/doi/10.1126/sciadv.adn0895
Log information:
Scientific progress